Biofilms in Canine UTIs: Why Some Infections Keep Coming Back
Biofilms in canine UTIs are one of the most important and least discussed reasons why some dogs stay stuck in the recurring infection cycle despite appropriate antibiotic treatment. Most conversations about canine urinary tract infections focus on bacteria — identification, antibiotics, clearance. But in dogs with persistent or frequently recurring infections, bacteria are only part of the story.
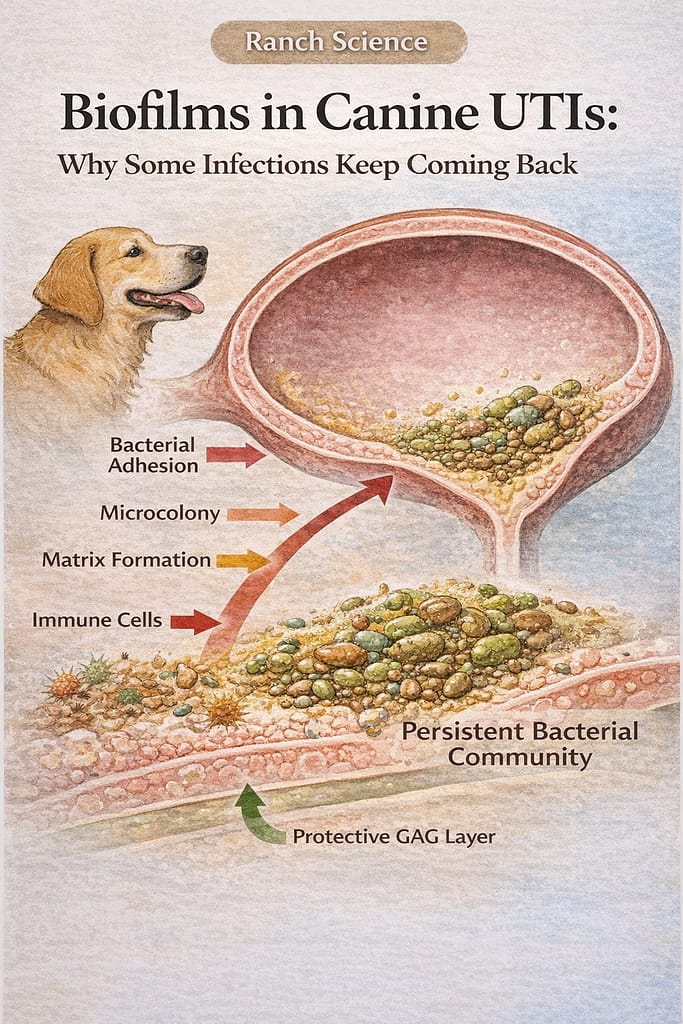
After initial adhesion to the bladder lining and activation of the immune response, certain bacterial populations can transition into a more organized and protected survival strategy: biofilm formation. These structured bacterial communities on the bladder surface help explain why some infections reappear despite treatment that appeared to work — and why the same antibiotic can become progressively less effective with each course.
What Is a Biofilm — And Why Does It Matter for Canine UTIs?
A biofilm is a structured community of microorganisms attached to a surface and embedded within a self-produced extracellular matrix. This matrix — made primarily of polysaccharides, proteins, lipids, and extracellular DNA — acts as a protective scaffolding that anchors the bacterial community to the bladder wall and shields it from external threats.
Inside a biofilm, bacteria behave fundamentally differently than free-floating (planktonic) cells. They communicate chemically through a process called quorum sensing, alter gene expression collectively, and shift into slower metabolic states that make them significantly less vulnerable to both antibiotics and immune cells. Rather than functioning as isolated organisms they operate as a coordinated community with collective defenses.
In the canine bladder the epithelial surface provides a substrate for biofilm attachment — particularly when the protective GAG layer has already been disrupted by previous infections or chronic inflammation. The more compromised the bladder surface, the more vulnerable it is to biofilm establishment.
How Biofilms Form in the Canine Bladder — Step by Step
Biofilm development in the bladder follows a predictable sequence that builds directly on the initial infection events.
Step 1 — Initial Adhesion: Bacteria attach to epithelial receptors on the bladder wall using fimbriae — hair-like surface projections. Adhesion becomes easier when the protective GAG layer is disrupted, exposing more binding sites on the underlying tissue. This is why dogs with a history of repeated infections are more vulnerable to biofilm formation than dogs experiencing their first UTI.
For the full science on how bacterial adhesion works: How Bacteria Adhere to the Bladder Wall in Dogs (and Why Recurring UTIs Keep Coming Back)
Step 2 — Microcolony Formation: Attached bacteria replicate and form small clusters. At this stage the community is still relatively vulnerable to antibiotics and immune cells — but this window closes quickly as the next steps progress.
Step 3 — Matrix Production: The developing community begins secreting extracellular polymeric substances — the protective matrix that gives biofilms their name and their resilience. This matrix anchors the community more securely to the bladder surface and begins limiting the penetration of antimicrobial agents.
Step 4 — Structural Maturation: The biofilm evolves into a three-dimensional architecture with internal channels that facilitate nutrient flow and waste exchange. At this stage the community is operating as a coordinated unit with collective chemical signaling, shared metabolic resources, and significant protection from external disruption.
Biofilms are not an immediate event — they are a progression. The earlier in this sequence that the bladder environment can be made less hospitable to bacterial adhesion and colonization, the lower the likelihood that mature biofilms establish.
The Inflammatory Environment and Biofilm Risk in Dogs
Inflammation is the body’s defense response to bacterial infection — but it also creates the surface conditions that make biofilm formation more likely. When bacteria attach to the bladder wall, the immune response triggers cytokine release, immune cell recruitment, and increased vascular permeability. These events fight the infection but also alter the epithelial tight junctions and disrupt the surface barrier.
When surface integrity declines as a result of inflammation, receptor exposure increases, adhesion becomes easier for subsequent bacteria, and structural instability persists even after the active infection resolves. This is why biofilm formation is more likely in a bladder that has already been inflamed by previous infections — the surface is already compromised when new bacteria arrive.
For the full science on what happens inside the bladder after bacterial adhesion: What Happens After Bacteria Stick? The Inflammatory Cascade Inside the Canine Bladder
This is why recurrence in dogs with a biofilm component is often not just about bacterial presence — it is about the surface condition of the bladder that bacteria encounter. A dog whose bladder surface has been repeatedly inflamed and incompletely recovered is a significantly more hospitable environment for biofilm establishment than a dog with intact surface integrity.
Why Biofilms in Canine UTIs Complicate Antibiotic Treatment
Biofilms don’t make bacteria invulnerable — but they significantly alter their responsiveness to treatment through several mechanisms operating simultaneously.
Reduced antimicrobial penetration: The extracellular matrix slows the diffusion of antibiotic molecules into deeper layers of the biofilm. Surface bacteria may be effectively killed while inner populations are exposed to sub-lethal concentrations — concentrations sufficient to create selection pressure for resistance but insufficient to clear the infection.
Altered metabolic states: Bacteria within mature biofilms often enter slower-growing or dormant phases. Most antibiotics are most effective against actively dividing bacteria — cells that are synthesizing new cell wall material or replicating DNA. Slow-growing biofilm bacteria are far less vulnerable to these mechanisms.
Community-based survival: Through quorum sensing — chemical signaling between bacteria — biofilm communities coordinate stress responses. When antibiotic pressure is detected, signaling can trigger community-wide shifts to more protective metabolic states. Individual bacteria in isolation cannot do this — it requires the organized community structure that only biofilms provide.
Physical immune evasion: Immune cells that would normally engulf and destroy free-floating bacteria struggle to penetrate the biofilm matrix. The three-dimensional structure physically limits immune cell access to bacteria in the deeper layers of the community.
These adaptations collectively explain why antibiotic courses that successfully clear planktonic bacterial populations may leave biofilm communities largely intact — and why infections with a biofilm component tend to recur more quickly and respond less well to repeated antibiotic courses over time.
For the full science on why antibiotics sometimes fail in recurring infections: Why Antibiotics Sometimes Fail in Recurring Dog UTIs
Persistence vs. Reinfection — Why the Distinction Matters
Not every recurring UTI involves biofilm formation. Understanding which recurrence pattern a dog is experiencing influences both the medical approach and the prevention strategy.
Reinfection — a genuinely new bacterial exposure from the external environment — is the most common cause of recurring UTIs. The original infection cleared completely but the dog’s anatomy, immune status, or daily environment creates repeated opportunities for new bacteria to ascend and establish.
Relapse — incomplete clearance of the original infection — occurs when antibiotic treatment suppresses bacterial populations enough to resolve symptoms but doesn’t fully eliminate them. The same bacteria re-establish from a residual population once treatment ends.
Biofilm-mediated persistence — the same bacteria reestablish from a structured surface community that survived the antibiotic course. This pattern typically shows a shorter interval between episodes with each recurrence, progressive reduction in antibiotic effectiveness, and symptoms that appear during or immediately after treatment ends.
Urine culture and sensitivity testing at each recurrence — combined with imaging to check for bladder stones that can harbor biofilms — helps distinguish between these patterns and guides appropriate management. Dogs with three or more UTIs in twelve months warrant this level of investigation rather than repeated empirical antibiotic prescribing.
The Role of the Bladder Environment in Biofilm Prevention
The bladder is not a passive container — it is an active barrier organ whose surface condition directly influences biofilm susceptibility. Several environmental factors affect whether the bladder presents a hospitable or inhospitable surface for biofilm development.
GAG layer integrity is the most critical surface factor. When this protective coating is intact it limits bacterial adhesion — the essential first step of biofilm formation. When it is compromised through repeated infection cycles more binding sites are exposed and the risk of biofilm establishment increases significantly. NAG and marshmallow root support GAG layer maintenance and recovery.
For the full science on the GAG layer: The Bladder’s Protective Barrier: Understanding the GAG Layer in Dogs
Hydration influences urine concentration and the osmotic stress on already inflamed bladder tissue. Adequate hydration doesn’t dissolve biofilms — but it maintains a urinary environment that is less favorable for the initial bacterial adhesion that biofilm formation depends on. Concentrated urine increases chemical irritation to the bladder surface and reduces the natural flushing that clears bacteria before they can establish.
For the full explanation of how hydration affects the urinary environment: Why Hydration Determines Whether Urinary Health Strategies Work in Dogs
Anti-adhesion daily support with cranberry PACs and D-Mannose addresses the adhesion step that initiates biofilm formation. By reducing the initial bacterial adhesion that leads to microcolony formation, these ingredients influence biofilm risk before the structured community can establish. This is why consistent daily use — not reactive dosing when symptoms appear — is essential for dogs prone to recurring infections.
What This Means Practically for Dogs With Recurring UTIs
For dog owners dealing with recurrent infections that seem to respond to antibiotics but keep coming back, the biofilm explanation has several practical implications.
Request urine culture and sensitivity testing at every recurrence — not just urinalysis. Culture identifies the specific organism and its antibiotic sensitivity pattern, which can reveal whether the same bacteria are recurring (suggesting persistence) or different bacteria are appearing (suggesting reinfection). Imaging to rule out bladder stones is essential — stones provide a non-biological surface for biofilm attachment that antibiotics cannot penetrate.
Support the bladder surface environment daily between infections. The window between episodes is where biofilm prevention is most actionable. A bladder surface supported by consistent hydration, daily anti-adhesion ingredients, and GAG layer support is significantly less hospitable to biofilm establishment than one receiving no daily support until symptoms appear.
For a complete daily prevention strategy: Dog UTI Prevention: Daily Habits That Actually Matter
For a practical evaluation of supplement formulas that support the bladder environment: Best Dog UTI Supplement: What to Actually Look For
Bladder Guard Soft Chews from Natural Ranch Products addresses the bladder environment daily — cranberry PACs and D-Mannose for anti-adhesion support, marshmallow root and NAG for GAG layer integrity, probiotics for immune function, and cold-pressed manufacturing to preserve the bioavailability of every active compound.
→ See Bladder Guard Soft Chews
→ See the Total Defense System
What is a biofilm in dogs?
A biofilm is a structured community of bacteria attached to a surface and embedded in a self-produced protective matrix made of polysaccharides, proteins, and extracellular DNA. In canine urinary infections biofilms form along the bladder wall — particularly when the protective GAG layer has been disrupted by previous infections. Inside the biofilm bacteria behave differently than free-floating cells, communicating chemically, shifting to slower metabolic states, and operating collectively in ways that significantly reduce their vulnerability to antibiotics and immune cells.
Can biofilms cause recurring UTIs in dogs?
Yes — biofilms are one of the mechanisms that contribute to recurring UTI patterns in dogs, particularly in cases where the same infection appears to resolve but returns quickly after antibiotic treatment ends. The biofilm matrix reduces antibiotic penetration, bacteria within biofilms shift to slower-growing states that antibiotics target less effectively, and the physical structure limits immune cell access. This allows bacterial communities to survive treatment courses that successfully clear free-floating bacteria.
Do antibiotics eliminate biofilms in dogs?
Antibiotics can reduce biofilm bacterial populations but often struggle to fully eliminate them. The extracellular matrix slows antibiotic penetration into deeper layers. Bacteria within mature biofilms shift to slower metabolic states that make them less vulnerable to antibiotics that target actively dividing cells. This is why infections with a biofilm component often recur more quickly and respond less well to repeated antibiotic courses — and why urine culture and sensitivity testing is essential for recurring infections rather than empirical antibiotic prescribing.
How do biofilms form in the dog bladder?
Biofilm formation follows a progression: bacteria first adhere to bladder wall receptors using fimbriae, then replicate into small clusters, then secrete extracellular matrix material that anchors and protects the community, and finally mature into a three-dimensional architecture with internal channels. The entire process is more likely when the bladder’s protective GAG layer has been compromised by previous infections — exposing more binding sites on the underlying tissue.
What is the difference between reinfection and biofilm persistence in dog UTIs?
Reinfection occurs when a new bacterial exposure from the external environment establishes after the previous infection fully cleared. Biofilm persistence occurs when a structured bacterial community survived the antibiotic treatment and re-establishes from those surviving populations. Distinguishing between these patterns requires urine culture at each recurrence — the same bacteria recurring in rapid succession suggests persistence while different bacteria or longer intervals between episodes suggest reinfection.
What reduces the risk of biofilm formation in dogs?
Reducing biofilm risk requires supporting the bladder environment at the adhesion step — before biofilms can establish. Consistent hydration maintains a less concentrated urinary environment less favorable for bacterial persistence. Daily anti-adhesion support with cranberry PACs and D-Mannose reduces the initial bacterial adhesion that biofilm formation depends on. GAG layer support with NAG and marshmallow root maintains the surface barrier that limits receptor exposure. These approaches work preventively and require consistent daily use between infections rather than reactive dosing when symptoms appear.
Scientific References
Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. “Urinary tract infections: epidemiology, mechanisms of infection and treatment options.” Nature Reviews Microbiology. 2015.
Hannan TJ, et al. “Host–pathogen checkpoints and population bottlenecks in persistent and intracellular uropathogenic E. coli bladder infection.” FEMS Microbiology Reviews. 2012.
Anderson GG, et al. “Intracellular bacterial biofilm-like pods in urinary tract infections.” Science. 2003.
Jacobsen SM, et al. “Complicated catheter-associated urinary tract infections due to biofilm-forming pathogens.” Clinical Microbiology Reviews. 2008.
Costerton JW, et al. “Bacterial biofilms: a common cause of persistent infections.” Science. 1999.
VCA Animal Hospitals. “Urinary Tract Infections (UTIs) in Dogs.” vcahospitals.com
